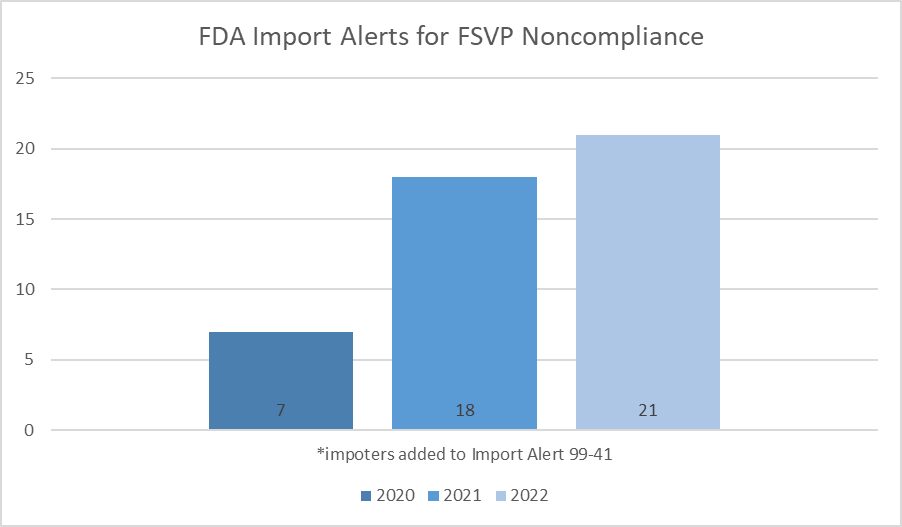
FDA continued to aggressively enforce the Foreign Supplier Verification Program (FSVP) in 2022 with an increase in Warning Letters, Import Alerts, and import refusals.
When looking behind the numbers, there continues the same general pattern: the importer failed two or three inspections and received a Form 483 each time, then they received a Warning Letter after either failing to respond to the 483 or responding insufficiently. The import alert listing then happened some time after FDA issued the Warning Letter. Of note, however, two importers were added to the import alert in 2022, but it appears that they did not receive a Warning Letter first.
2022 witness a dramatic increase in import refusals — a 465% increase! Going from 114 refused lines to 644. The obvious take away: FDA will refuse an importer’s shipments if they do not comply with FSVP.
Click here for a refresher on the FSVP basics.